NKF KDOQI GUIDELINES
KDOQI Clinical Practice Guidelines for Bone Metabolism and Disease in Chronic Kidney Disease
GUIDELINE 4. RESTRICTION OF DIETARY PHOSPHORUS IN PATIENTS WITH CKD
4.1 Dietary phosphorus should be restricted to 800 to 1,000 mg/day (adjusted for dietary protein needs) when the serum phosphorus levels are elevated (>4.6 mg/dL [1.49 mmol/L]) at Stages 3 and 4 of CKD, (OPINION) and >5.5 mg/dL (1.78 mmol/L) in those with kidney failure (Stage 5). (EVIDENCE)
4.2 Dietary phosphorus should be restricted to 800 to 1,000 mg/day (adjusted to dietary protein needs) when the plasma levels of intact PTH are elevated above target range of the CKD stage (see Table 15 in Guideline 1). (EVIDENCE)
4.3 The serum phosphorus levels should be monitored every month following the initiation of dietary phosphorus restriction. (OPINION)
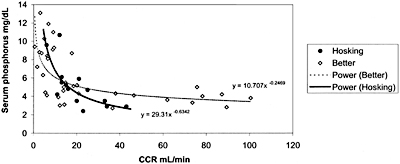
Fig 9. Relationship between serum phosphorus and CCR.
Background
Levels of serum phosphorus in patients with CKD remain within the normal range or may even be modestly below the normal range until the GFR declines to 20 to 30 mL/min/1.73 m2 (Stage 4 of CKD). At these latter levels of GFR, hyperphosphatemia becomes evident (Fig 9). Therefore, it may appear that dietary phosphate intervention is not necessary in patients with Stages 1, 2, and 3 of CKD. However, phosphate retention occurs very early in the course of CKD (probably at Stage 1, but definitely at Stage 2) and such phosphate retention participates in the genesis of secondary hyperparathyroidism. Indeed, the blood levels of PTH are elevated when GFR falls to 60 mL/min/1.73 m2, even though serum phosphorus levels are not elevated. Studies in animals85,101 and in human adult patients102 and children showed that dietary phosphate restriction in proportion to the decrease in GFR in the early and moderate stages of CKD (Stages 2 and 3) when blood levels of PTH are elevated and serum phosphorus levels are normal, was effective in lowering blood PTH levels.
Rationale
Since phosphate retention occurs early in the course of CKD (probably Stage 1, but certainly Stage 2) and contributes to the genesis of secondary hyperparathyroidism, and since PTH levels begin to rise when GFR falls below 60 mL/min/1.73 m2 (Stage 3 of CKD), even when serum phosphorus levels are normal, the plasma level of PTH is a better marker in the early course of CKD for the need to begin dietary phosphate restriction than is serum phosphorus, calcium, or creatinine levels. Of course, at later stages of CKD (Stage 4 and 5), the levels of serum phosphorus are elevated and therefore dictate the need for the prescription of dietary phosphate restriction, as an essential approach to controlling serum phosphorus and plasma levels of intact PTH.
Strength of Evidence
The efficacy of dietary phosphate restriction and the timing of its initiation were assessed through an evidence-based review of the literature. Because dietary phosphate restriction alone is most commonly used in CKD patients, the focus was on the following questions:
(1) What is the correlation between serum phosphate levels and outcomes?
(2) How effective is dietary phosphate restriction at different stages of CKD?
(3) What percentage of patients comply with dietary phosphate restriction and how does noncompliance affect patient outcomes?
(4) What side-effects are associated with a phosphorus-restricted diet?
Unfortunately, there were no studies in CKD patients that met the inclusion criteria and addressed the relationship between serum phosphate and multiple indices of morbidity and mortality such that an absolute level of serum phosphorus could be used to guide initiation of dietary phosphate restriction.
The relationship between kidney function and serum phosphorus was evaluated. The included studies evaluated the relationship between serum phosphorus and creatinine clearance or serum creatinine (Cr)103-105; they showed an inverse relationship between serum phosphorus levels and kidney function. Two studies104,106 that could be used to determine the correlation between serum phosphorus levels and creatinine clearance reported data on 51 patients (Fig 9). These studies showed that serum phosphorus levels begin to rise when creatinine clearance falls below 20 to 30 mL/min/1.73 m2 (0.33 to 0.50 mL/s/1.73 m2). Other studies on 121 patients reporting a direct relationship between serum phosphorus and serum Cr showed no clear indication as to the absolute level of serum Cr that is associated with hyperphosphatemia.103-108 Taken together, the data support an association between the decline in kidney function and the rise in serum phosphorus, with increments in the latter becoming evident when creatinine clearance falls to 20 to 30 mL/min/1.73 m2 (0.33 to 0.50 mL/s/1.73 m2) (Stage 4).
However, there are other confounding issues when utilizing the serum Cr or creatinine clearance as a guide for the initiation of dietary phosphorus restriction, since serum creatinine is influenced by existing muscle mass, which varies from patient to patient. There may be patients who have increased levels of serum phosphorus at a lower level of serum creatinine because of their lower muscle mass. Most importantly, a rise in the serum phosphorus level represents a failure of the normal compensatory response of elevated parathyroid hormone to adequately increase kidney excretion of phosphate. It appears that at creatinine clearances of 20 to 30 mL/min/1.73 m2 (0.33 to 0.50 mL/s/1.73 m2), the maximum decrement in kidney reabsorption of phosphate has been reached. With further deterioration in kidney function, there is no further increase in urinary excretion of phosphate, even in the presence of elevated parathyroid hormone; thus, serum phosphorus levels begin to rise. There are no reports that met the inclusion criteria to specifically identify the level of kidney function at which the decrement in reabsorption of phosphate by the kidney reaches its maximum and/or at what levels of serum phosphorus, in the course of CKD, blood PTH levels begin to rise.
In summary, the available data and the opinion of the Work Group support the proposal that dietary phosphate restriction should be initiated when blood PTH levels begin to rise (Stage 2) and/or when serum phosphorus levels are elevated at any stage of CKD.
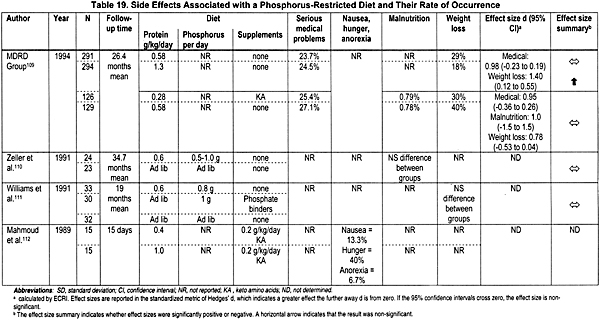
The effectiveness of dietary phosphate restriction in controlling the hyperphosphatemia of CKD was analyzed in 19 studies examining 2,476 patients. Fifteen randomized controlled trials110-124 and 4 nonrandomized controlled trials125-128 met the inclusion criteria. The last 4 studies evaluated the side effects of dietary phosphate restriction (Table 19). The vast majority of studies evaluated restricted protein diets, which are usually (but not always) equivalent to low phosphorus diets. In many of these studies, calcium100,103-108,129,130 and vitamin D supplements were used,87,90,92,93,100,103-108,129-131 or phosphate binders were also administered to the patients in addition to the dietary intervention. Thus, the interpretation of these data should be done with caution. Various end-points were utilized:
- Quality of life. One study reported that low protein diet did not adversely affect employment,127 but the results of the Modification of Diet in Renal Disease (MDRD) study indicated that patients with CKD (Stages 3 and 4) treated with very restricted protein diets were less able to socialize.132
- Mortality. Nearly all of the included studies evaluated the role of dietary restriction of protein/phosphorus on mortality. The reported results were variable. When these data were analyzed by meta-analysis, no effect on mortality was found.
- Kidney Function. Seven of nine studies in adult patients with CKD suggest that dietary phosphorus restriction may stabilize kidney function.110,111,113,116,117,121,123,125,128 Conclusions in this regard could not be drawn from studies in children or in adults with severe CKD.
- Bone and Mineral Metabolism. Several small studies reported that dietary phosphorus restriction in patients with CKD had no significant effect on serum alkaline phosphatase,112,115,117,126 PTH levels112,117,125,127 serum calcium levels,112,115,117,125,126 serum phosphorus levels,112,115,122, 125,126,133 and urinary phosphate excretion.111,119,122 In contrast, in a careful and well-controlled study of 4 patients with Stages 1 and 2 of CKD conducted in a metabolic ward before and after 8 weeks of dietary phosphate restrictions in proportion to the decrement in GFR, there was a reduction in blood PTH levels to normal without significant changes in the serum levels of phosphorus, significant decrements in blood levels of alkaline phosphatase and in urinary excretion of phosphate, and significant increments in blood levels of 1,25(OH)2D3 and intestinal absorption of calcium.10 Also, the dietary phosphate restriction was associated with marked improvement in bone resorption and defects in bone mineralization as evidenced by studies of bone biopsy.10
Moderate dietary phosphate restriction is also appropriate for children, provided close monitoring of linear bone growth is exercised, as no study provided evidence for adverse effects.116,120,134,135 In addition, studies in adults did not support any adverse effect on nutritional status as a result of dietary phosphate restriction.110-112,115-117,119,123,126,128,136,137
Compliance. Compliance with dietary restriction in the research setting of clinical studies may not reflect the situation in clinical practice. While compliance with dietary phosphorus restriction in clinical practice is commonly believed to be poor, there is a lack of data to support this supposition. Most studies have found compliance rates of 35% to 91% with low-protein diets.102,116,122,132 One study reported 41% and 77% compliance at years 1 and 3, respectively.138 The compliance rates with dietary phosphate restriction were similar to compliance rates for low-protein diets. It was not addressed whether the improvement at year 3 is related to continuous education and/or the realization by the patient of the adverse effects of noncompliance.
Given the lack of evidence of adverse effects, and the evidence of positive benefit of dietary phosphate restriction, it is the consensus of the Work Group that dietary phosphate restriction be initiated in patients with CKD when PTH levels are elevated (GFR <60 mL/min/1.73 m2, Stage 3) or with elevated blood levels of serum phosphorus (Stages 4 and 5).
Limitations
Despite the relatively large number of prospective randomized trials evaluating dietary phosphorus restriction, most of these studies specifically utilized protein-restricted diet and therefore indirectly restricted phosphate intake. While protein and phosphorus are closely related in foods, it is possible to restrict protein without fully restricting phosphorus. Much of the data is also difficult to interpret since most of the reports provided analysis for "prescribed diet" rather than "consumed diet." Furthermore, in many studies, the patients had concomitant therapy with Vitamin D and/or phosphate binders making interpretation of the results difficult.
While the available data do not support the common belief that dietary phosphate restriction negatively impacts nutritional status, it must be stressed that dietary phosphate restriction has the potential of adversely impacting nutritional status if done in a haphazard manner. The data that demonstrate the ability to maintain good or stable nutritional status during dietary phosphate restriction were obtained in studies in which dietitians provided careful instruction and regular counseling and monitoring. In the research setting, patients are monitored closely and have regular contact with their kidney-care providers. Those patients who have been "casually" instructed to watch their protein or phosphate intake, without regular follow-up, may be at risk for serious side-effects such as malnutrition. Unfortunately, there are no data on those patients who are not regularly and closely followed.
Clinical Applications
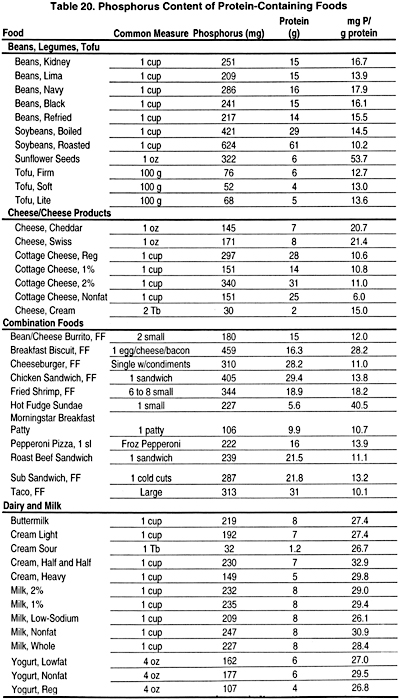
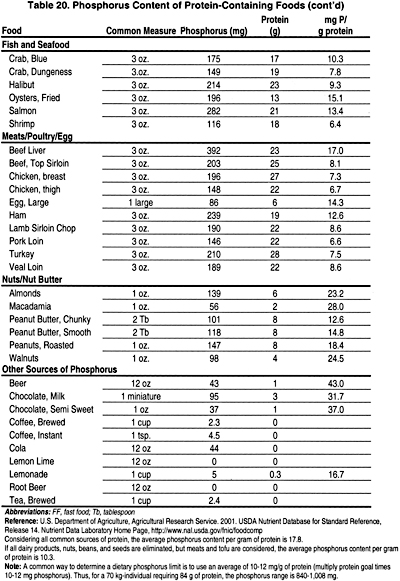
It is critical to provide consistent instruction and regular follow-up during prescription of dietary phosphate restriction. In patients with early stages of CKD (Stages 2 and 3), compliance with dietary phosphate restriction is difficult and requires intensive dietitian support. In patients with advanced CKD (Stages 4 and 5), restriction of dietary phosphorus is more easily accomplished because of concomitant dietary protein modification. However, in CKD patients treated with dialysis (Stages 5), care must be taken to reduce phosphate intake while maintaining adequate protein intake as recommended by the KDOQI Guidelines on Nutrition (www. kdoqi.org). In individuals >80 kg, it is impossible to plan a palatable diet with adequate protein while limiting the phosphate intake to <1,000 mg. In view of this limitation, the phosphate level of the diet should be as low as possible while ensuring an adequate protein intake. If one multiplies the recommended protein level times 10 to 12 mg phosphate per gram of protein, a reasonable phosphate level can be estimated. The average amount of phosphorus per gram of protein ranges from 12 to 16 mg. In order to limit phosphorus significantly, those protein sources with the least amount of phosphorus must be prescribed (Table 20). There is clear need for more active involvement and reimbursement for counseling from renal dietitians in the care of patients with CKD, beginning with Stage 2. This enhanced involvement has been hindered by inadequate reimbursement for the services of qualified renal dietitians. While the Medical Nutrition Act of 2002 funds nutrition services only to Medicare-eligible patients, it does not provide for all CKD patients who need ongoing nutrition services prior to the need for dialysis therapy.
Recommendations for Research
There is a need for large, multi-center longitudinal studies evaluating the effects of dietary phosphate restriction (as opposed to only protein restriction) on nutritional status, growth in children, morbidity, mortality, bone disease, and progression of decline in kidney function. These studies should be conducted in patients with all stages of CKD beginning in Stage 2.