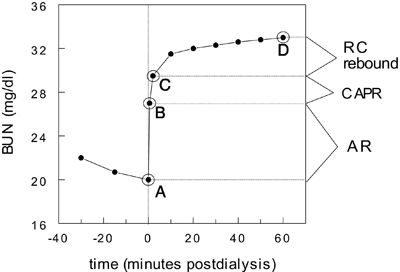
Fig 3. Components of postdialysis urea (BUN) rebound. See text for explanation. Reprinted with permission.82
When dialysis adequacy is assessed by using predialysis and postdialysis BUN measurements, blood samples should be drawn by using certain acceptable procedures.
3.1 Both samples (predialysis and postdialysis) should be drawn during the same treatment session. (A)
3.2 The risk of underestimating predialysis BUN level because of saline dilution or by sampling the blood after treatment has begun should be avoided. (A)
3.3 The risk of underestimating the postdialysis BUN level because of access recirculation (AR) should be avoided by first slowing the blood flow through the dialyzer to a rate at which AR is expected to be minimal (100 mL/min) for a period long enough to ensure that unrecirculated blood has advanced to below the sampling port (usually 15 seconds). (A)
3.4 An alternative method is to stop the dialysate flow for a period long enough to increase the dialysate outlet BUN level close to that of the blood inlet BUN level (3 minutes) before obtaining the postdialysis sample. (A)
Summary of Updated Changes
The proper methods of sampling blood for urea nitrogen before and after an HD treatment were detailed in Guidelines 7 through 9 of the previously published KDOQI 2000 HD Adequacy Guidelines.6 These updated guidelines, 3.1, 3.2, and 3.3, are largely unchanged from the 2000 guidelines, except for minor details. When sampling blood from venous catheters, the volume of the initial aspirate is specified more precisely, and a recommendation is made to discard—instead of routinely reinfusing—the aspirated blood sample. Guideline 3.4, acknowledging the alternative use of the dialysate-stop-flow method, is new.
As reviewed in the 2000 guidelines,6 there are 3 components of postdialysis urea nitrogen rebound (see Fig 3). The first is caused by AR, which resolves within seconds after stopping dialysis (point B), the second is caused by CAPR, which resolves within 1 to 2 minutes after stopping dialysis (point C), and the third is caused by entry of urea from relatively undialyzed tissues and body compartments, which we term remote-compartment (RC) rebound. The latter resolves within 30 to 60 minutes after stopping dialysis (point D).

Fig 3. Components of postdialysis urea (BUN) rebound. See text for explanation. Reprinted with permission.82
The first focus of these blood-drawing guidelines is to limit the effect of AR on the postdialysis BUN sample because AR causes large overestimations of the true delivered dose and can result in true delivered Kt/V values less than 0.8 (at which level mortality risk is strongly increased) in patients with apparent Kt/V values of 1.4 or greater.83 Since the KDOQI 2000 guidelines were published, it has become clear that the later rebound caused by CAPR is small84 and effects of RC rebound are relatively predictable based on the rate of dialysis.84,85 In addition, some studies showed that sampling blood about 30 minutes before the end of dialysis can predict the BUN level 30 minutes after the end of dialysis.86 This method is not recommended in adults because of its relative complexity and because RC rebound is relatively predictable based on the rate of dialysis,84,85 and—most importantly—because in the presence of AR, the dialysis dose can still be markedly underestimated unless a slow-flow method is used to draw the sample 30 minutes before the end of dialysis.
Predialysis Blood Sampling Procedure (CPG 3.1 and 3.2, see Table 5)
The predialysis BUN sample must be drawn before dialysis is started to prevent this sample from reflecting any impact of dialysis. Dilution of the predialysis sample with saline or heparin must be avoided. Underestimating the predialysis BUN level will result in underestimation of delivered Kt/V or URR, which is not particularly dangerous; however, nPCR then will be underestimated.
Postdialysis Blood-Sampling Procedure (CPG 3.3, see Table 6)
Proper timing for acquisition of the postdialysis BUN sample is critical.6,83 Immediately upon completion of HD, if AR was present, some of the blood remaining in the access and extracorporeal circuit actually is recirculated blood. If the blood sample is drawn immediately upon completion of dialysis, just-dialyzed blood that has recirculated into the access will dilute the sample. The consequence of sampling this admixture is a falsely decreased BUN value and artificially elevated Kt/V and URR.6,83 Therefore, the amount of dialysis delivered will be overestimated.
Early urea rebound (≤3 minutes after dialysis) may be viewed as a 2-component process.89-91 The first component is caused by blood recirculation within the access or catheter and is not present in patients without AR. If AR is present, urea rebound from recirculation begins immediately upon completion of HD and resolves in less than 1 minute, usually within 20 seconds. The second component of early urea rebound is caused by CAPR that begins approximately 20 seconds after stopping HD and is completed 2 to 3 minutes after slowing or stopping the blood pump.90 CAPR refers to the routing of just-dialyzed blood through the veins to the heart, through the pulmonary circuit, and back to the access without the passage of the just-dialyzed blood through any urea-rich tissues.90-93 It should not occur with a venous access because venous (rather than arterial) blood is sampled; however, some increase in urea concentration during the initial 3-minute time frame may occur because of mixing of urea returning from different organs. The late phase of urea rebound (>3 minutes) is completed within 30 to 60 minutes after the cessation of dialysis. The late phase is a consequence of flow-volume disequilibrium (perfusion or parallel-flow model)94 and/or delayed transcellular movement of urea (diffusion model)92,95 (see CPG 2, Methods of Measuring and Expressing the HD Dose).
Why the Blood Pump Should Be Slowed Before Sampling
Decreasing blood flow to 100 mL/min reduces the entry of cleared blood into the access and stops AR (unless there is needle reversal, in which case it still greatly reduces AR). The dead space of the blood tubing attached to the access needle usually is about 3 mL. The dead space in most venous catheters is similar, albeit somewhat less, in the range of 1 to 2 mL. The dead space between the tip of the dialyzer inlet (arterial) blood tubing and sampling port area usually is about 7 to 12 mL, giving a total dead space of 10 to 15 mL, although this should always be measured and known for a given set of blood tubing because in some blood tubing, the sampling port is farther removed from the patient connection. A flow rate of 100 mL/min is about 1.6 mL/s. Therefore, waiting 15 seconds at such a flow rate will ensure that the column of undiluted blood will have moved 1.6 × 15 = 24 mL into the blood tubing during the time of reduced blood flow. As long as the volume of tubing between the patient connection and sampling site is substantially less than this 24 mL, the sampled blood should not be contaminated with outflow blood.
In situations in which the blood is drawn not from a sampling port on the inflow blood tubing, but by attaching a Luer-Lock connector (Becton Dickinson and Co., Franklin Lakes, NJ, USA) to either the venous catheter or arterial needle blood tubing, the dead-space volume to the sampling site is only 2 to 3 mL. However, for simplicity, the Work Group recommended keeping the slow-flow period the same regardless of the site from which blood is sampled.
Stopping Dialysate Flow Before Sampling
A new method for postdialysis blood sampling introduced since the KDOQI 2000 Guideline update was reviewed by the Work Group.96,97 When dialysate flow is stopped, the dialysate outlet urea concentration starts to approach the blood inlet level, and AR (if present) has a progressively lower dilutional effect on inlet blood flow. With this method, as outlined in Table 7, blood flow must not be reduced because the dialysate, now “trapped” in the dialyzer, needs to equilibrate with blood as quickly as possible. Two studies showed that 5 minutes was adequate to equilibrate the arterial and venous blood tubing samples.96,97 The Work Group recommendation is to follow the method of Geddes et al.96 It should be realized that 3 minutes after stopping dialysis, the CAPR component of rebound will be complete and RC rebound will have begun. Hence, a postdialysis BUN sample drawn by using this dialysate method will be slightly higher than that obtained when using the blood method because with the latter, the sample is obtained only 15 seconds after the end of dialysis. This means that the spKt/V obtained by using the stop-dialysate-flow method will (theoretically) be slightly lower in comparison to the slow-the-blood-flow method.
Use of a 5-minute waiting period resulted in a 2% decrease in measured value for URR (Fig 4).96 Because of the rebound considerations discussed and based on data in Fig 4, the Work Group decided that a 3-minute waiting period was sufficient. By that time, dialyzer inlet and outlet samples have nearly equilibrated.
Fig 4. Stop-dialysate method for postdialysis blood sampling. Mean arterial and venous blood urea concentrations after stopping dialysate flow are expressed as a fraction of the blood urea concentration in the contralateral arm at time zero (n = 10). The data suggest that, for practical purposes, 3 minutes after stopping dialysate flow, equilibration has occurred between inlet and outlet blood. Reprinted with permission.96
The stop-dialysate-flow method has not been validated during pediatric dialysis. If a large dialyzer is used at a relatively lower blood flow rate, the dialyzer outlet blood may still have a substantially lower urea concentration than inlet blood after 3 minutes of stopping dialysate flow.