The Hb target is the intended aim of ESA therapy for the individual patient with CKD. In clinical practice, achieved Hb results vary considerably from the Hb target.
2.1.1 In the opinion of the Work Group, selection of the Hb target and selection of the Hb level at which ESA therapy is initiated in the individual patient should include consideration of potential benefits (including improvement in quality of life and avoidance of transfusion) and potential harms (including the risk of life threatening adverse events). (Clinical Practice RECOMMENDATION)
2.1.2 In the opinion of the Work Group, in dialysis and nondialysis patients with CKD receiving ESA therapy, the selected Hb target should generally be in the range of 11.0 to 12.0 g/dL. (Clinical Practice RECOMMENDATION)
2.1.3 In dialysis and nondialysis patients with CKD receiving ESA therapy, the Hb target should not be greater than 13.0 g/dL. (Clinical Practice GUIDELINE - MODERATELY STRONG EVIDENCE)
KDOQI CPGs and CPRs for Anemia in CKD, published in May 2006, included recommendations for Hb targets that were based on a systematic review and structured appraisal of RCTs comparing treatment to different Hb targets. After publication of these guidelines, 5 additional RCTs were published.1-5 An additional small trial published in 2005 was unintentionally omitted in the previous evidence review.6 The new studies expanded the evidence on clinically important outcomes, doubled the number of all patients with CKD examined, and increased the number of nondialysis patients with CKD studied in RCTs from 575 to 3,432. In keeping with criteria for updating a systematic review and guidelines before a scheduled revision (Methods), the Work Group undertook a reexamination of the available evidence on Hb treatment targets. The reexamination included the new studies,the study not included in the previous review, and those appraised previously.
Although new evidence relevant to other topics covered in the KDOQI CPGs and CPRs for Anemia in CKD published in May 2006 is also continuously accumulating, the Work Group concluded that the evidence published since the last review and relevant to CPGs and CPRs 1.1 (Identifying Patients and Initiating Evaluation), 1.2 (Evaluation of Anemia in CKD), 3.1 (Using ESAs), 3.2 (Using Iron Agents), 3.3 (Using Pharmacological and Nonpharmacological Adjuvants to ESA Treatment in Hemodialysis-CKD, 3.4 (Transfusion Therapy), and 3.5 (Evaluating and Correcting Persistent Failure to Reach or Maintain Intended Hb) does not meet criteria for an update. For these CPRs and CPGs, for corresponding CPRs in children with anemia, and for anemia in patients after kidney transplantation, the reader is referred to the May 2006 document. 56
The updated CPRs (2.1.1 and 2.1.2) and CPG (2.1.3) are intended to assist the practitioner caring for patients in selecting Hb targets appropriate for individual patients receiving ESA therapy or considered for ESA therapy, whether or not they are also receiving iron therapy. Recommended Hb targets apply exclusively to patients receiving ESA. Hb targets are not intended to apply to the treatment of iron deficiency in patients receiving iron therapy without the use of ESAs.
Warnings, indications, precautions, and instructions for dosing and administration of ESAs are available from national regulatory agencies, including the United States Food and Drug Administration (FDA), and product package inserts.7-9 The Work Group directed considerable thought and attention in particular to the most recent FDA-approved prescribing information. Appendix 1 provides a detailed comparison of KDOQI CPGs and CPRs (May 2006 and update 2007) with FDA-approved prescribing information current as of March 2007.
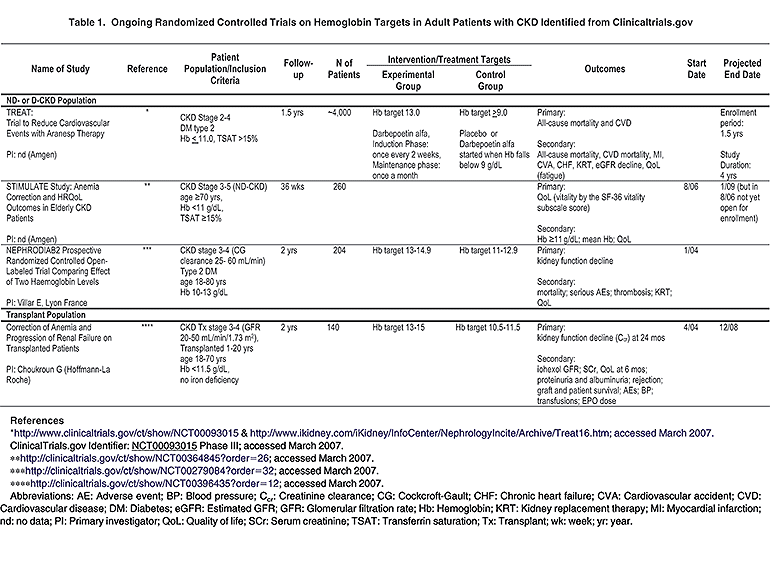
Ongoing and future trials in patients with CKD are expected to provide more information on ESA use and Hb targets, including treatment with ESAs compared with placebo and higher compared with lower Hb targets (Table 1).

Selection of the Hb target and selection of the Hb level at which ESA therapy is initiated in the individual patient should include consideration of potential benefits (including improvement in quality of life and avoidance of transfusion) and potential harms (including the risk of life-threatening adverse events).
The Work Group chose the wording, order, and placement of this statement to guide practitioners in selecting a Hb target for ESA therapy and a Hb level at which ESA therapy is initiated in the individual patient with CKD and anemia. The statement reflects the conclusion that improvement in quality of life and avoidance of transfusion are the most likely treatment benefits and that there is potential for harm when aiming for high Hb targets.
The statement selection of the Hb target and selection of the Hb level at which ESA therapy is initiated in the individual patient captures the conclusion of the Work Group that the selection of the Hb target for ESA therapy and the selection of the Hb level at which ESA therapy is initiated in the individual patient are separate but related steps in medical decision making for the individual patient. In available RCTs, treatment has generally been initiated when the baseline Hb decreases within, at, or less than the assigned Hb target.
The statement should include consideration reflects the limitations of our current evidence base, which does not allow precise recommendations for each individual patient (see the section Limitations of Evidence). The statement also acknowledges that judgments about benefits and harm may vary from patient to patient and for the same patient under different conditions. Limitations of the current evidence base, differences in individual judgments, and variable responsiveness between patients and within a patient argue for engaging the patient and for maintaining flexibility when setting Hb targets for ESA therapy.
Reference to quality-of-life benefit reflects the appraisal that when selecting the Hb target, an improvement in quality of life should be an expected treatment benefit. Quality of life is an outcome of direct importance to patients and should be valued accordingly.10 Although health related quality of life (HRQoL) is not usually quantified in a systematic fashion in clinical practice, in research studies, measurement of HRQoL is performed by using standardized instruments that have been validated in a range of target populations, including patients with CKD requiring or not requiring dialysis. Results yielded by these instruments achieve levels of reliability and precision that are comparable to those seen with other commonly used clinical tests.11 HRQoL has been examined in several RCTs comparing lower and higher Hb targets in patients with CKD receiving ESAs for anemia. Although it is difficult to aggregate HRQoL effects across studies because different HRQoL instruments were used and some reports lacked detail, low-quality evidence suggests benefit of HRQoL (Tables 3 and 9):
Reference to avoidance of transfusions reflects the appraisal that higher compared with lower Hb targets are associated with a decrease in red blood cell transfusion rates in hemodialysis patients (Table 2). Assignment to Hb targets greater than 13 g/dL decreases, but does not eliminate, transfusions in hemodialysis patients.14 Transfusion-related risks are discussed in detail elsewhere (CPR 3.456).
Potential harms refers to evidence from RCTs suggesting that assignment to Hb targets greater than 13.0 g/dL may increase the risk of lifethreatening adverse events. This evidence is discussed in detail in the rationale to statement 2.1.3.
The distinction between Hb targets and achieved Hb levels is fundamental to the development of this guideline. In considering information available to guide selection of Hb targets, we specifically excluded evidence from Hb levels achieved in RCTs or reported in observational studies. Whereas higher achieved Hb levels in patients assigned to similar target Hb levels is associated with decreased risk of mortality and hospitalization,12,14,16-20 treatment assignment to target Hb levels greater than 13.0 g/dL may increase the risk of life-threatening cardiovascular events.
The consensus opinion of the Work Group that potential benefits, for improvement in HRQoL and avoidance of transfusion, and potential harms must each be considered, coupled with an absence of specific quantitative information to assist the practitioner in weighing each component, renders statement 2.1.1 a CPR.
In dialysis and nondialysis patients with CKD receiving ESA therapy, the selected Hb target should generally be in the range of 11.0 to 12.0 g/dL.
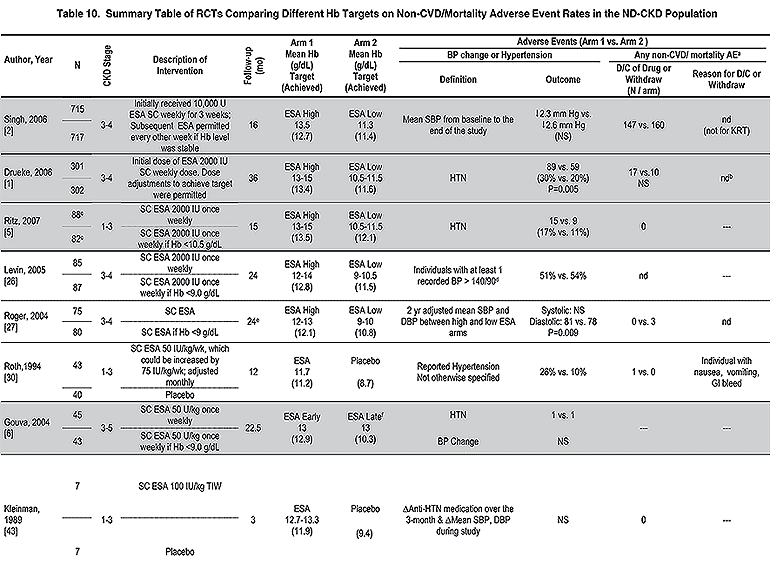
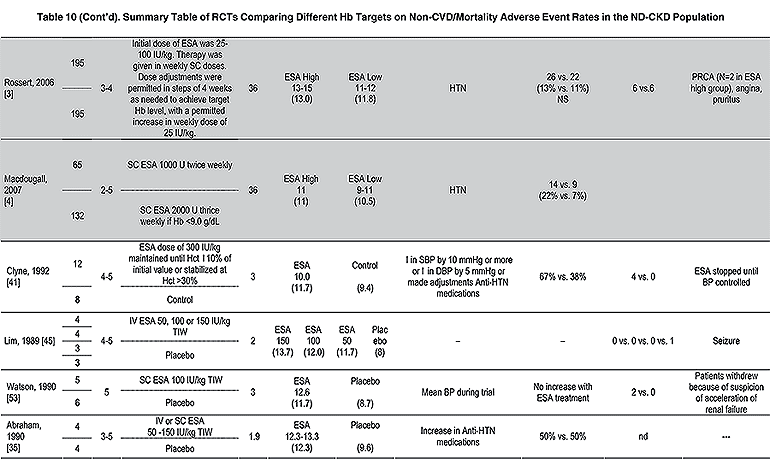
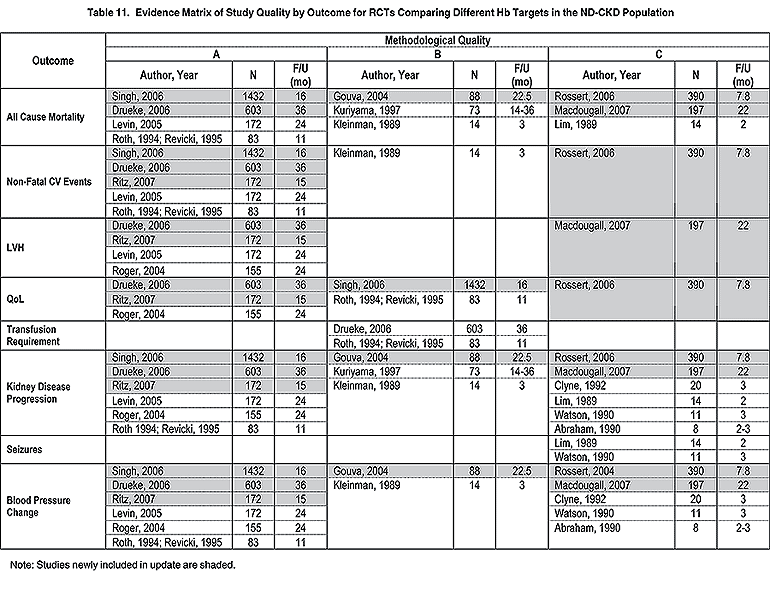
Evidence supporting the statement that in dialysis and nondialysis patients with CKD receiving ESA therapy, the selected Hb target should generally be in the range of 11.0 to 12.0 g/dL includes results from 14 RCTs in dialysis patients and 15 RCTs in nondialysis patients and is presented in detail for each trial (Tables 2 to 5 and 8 to 10) and in summary for each outcome (Tables 6, 7, 11, and 12).
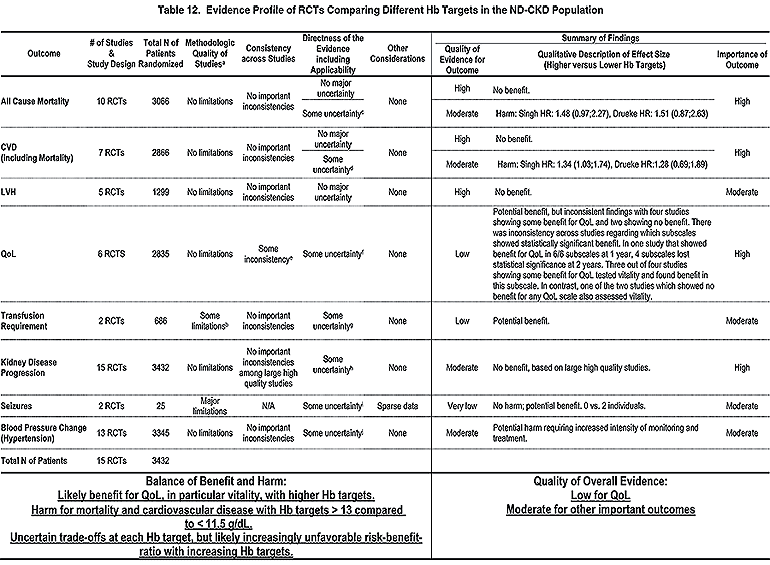
The evidence considered by the Work Group to support the statement is confined to results of between-group comparisons generated by intention-to-treat trials that randomly assigned patients to distinct Hb targets, including trials that used ESAs in both treatment arms and trials that used ESAs in 1 treatment arm and either placebo or no treatment in the control arm (Fig 1).
The practitioner approaches the decision to select a Hb treatment goal with the intent to treat the individual patient and should expect that the achieved Hb level will vary considerably from the intended Hb target. To develop these guidelines and recommendations, we therefore appraised only evidence that was generated from intent-to-treat analyses of trials in patients randomly assigned to either higher or lower Hb targets.
The evidence base for the statement the selected Hb target should generally be in the range of 11.0 to 12.0 g/dL includes results from trials that examined Hb targets from 6 to 16 g/dL (Tables 2 to 5 and 8 to 10; Fig 1). Early RCTs differ substantially from later RCTs in both size and Hb targets. RCTs conducted before 1998 are characterized by smaller study size, upper Hb targets in the range of 10 to 13 g/dL, and lower Hb targets that reflect assignment to placebo or no-treatment control. Trials published in 1998 and thereafter are characterized by larger study size, higher Hb targets in the range of 12 to 16 g/dL, and lower Hb targets between 9 and 12 g/dL. In more recent trials, by comparison, Hb baseline values are higher than those seen in early trials. Moreover, recent RCTs set lower targets at Hb levels well above those in earlier trials, in which patients in the control arm were assigned to placebo or no-treatment control groups. Both effects combine to render differences between Hb targets smaller in more recent trials.
In the statement the selected Hb target should generally be in the range of 11.0 to 12.0 g/dL, the Work Group used the word target to distinguish between a Hb target and an achieved Hb level. In hemodialysis patients receiving ESA therapy with a Hb target in the range of 11.0 to 12.0 g/dL, the proportion of patients who achieve Hb levels from 11.0 to 12.0 g/dL in a single month may be 30% or less.21,22 Moreover, achievement of a Hb level within the 11.0- to 12.0-g/dL target in hemodialysis patients is transitory. More than 90% of patients experience cyclical Hb excursions averaging 10.3 weeks in duration and 2.5 g/dL in amplitude. 23 In part because of these fluctuations, approximately 50% of patients who achieve a Hb level in a 11.0- to 12.0-g/dL target range in 1 month will show Hb results greater or less than that range in the subsequent month.21,22 Given the variability in Hb levels observed in clinical practice, the width of a Hb interval that would encompass 95% of Hb results in a population of dialysis patients undergoing ESA therapy could be as high as 5.6 g/dL.24 Accordingly, to ensure that no more than 2.5% of patients exceed a Hb target of 12.0 g/dL, a target range designed to include 95% of patients would have a lower Hb limit of 6.4 (that is, 12.0 minus 5.6) g/dL.

In the statement the selected Hb target should generally be in the range of 11.0 to 12.0 g/dL, the word generally emphasizes the need to maintain flexibility in medical decision making given the breadth of variability between patients' individual needs, values, functional status, disease burden, prognosis, and responsiveness to ESA therapy (Rationale for CPR 2.1.1).
In the statement the selected Hb target should generally be in the range of 11.0 to 12.0 g/dL, the 2 specific values 11.0 g/dL and 12.0 g/dL define inclusively either a single Hb target range (11.0 to 12.0 g/dL) or a range of possible single-point Hb targets between 11.0 and 12.0 g/dL; entail unavoidable subjectivity in selecting Hb cutoff values; explicitly exclude reference to achieved Hb levels; and together reflect the efforts of the Work Group to balance the potential quality-of life benefits and avoidance of transfusion gained by ESA therapy against the potential harm suffered by patients with Hb targets greater than 13 g/dL.
Available RCTs illustrate the distinction between a Hb target range1,3,5,26,27 and a discrete Hb target (Fig 1).2,14
The lack of information to support specific Hb cutoff values in defining an optimum Hb target renders statement 2.1.2 a CPR.

In dialysis and nondialysis patients with CKD receiving ESA therapy, the Hb target should not be greater than 13.0 g/dL.
The conclusion that the Hb target should not be greater than 13.0 g/dL is based on analysis of all-cause mortality and adverse cardiovascular events in patients with CKD assigned to Hb targets greater than 13.0 g/dL compared with lower Hb targets for ESA therapy (Tables 2, 4, 6 to 8, and 10 to 12). These trials evaluated whether a Hb target greater than 13.0 g/dL would prevent adverse cardiovascular events or mortality, testing the specific hypothesis that rates of fatal and nonfatal cardiovascular events or all-cause mortality in patients assigned to Hb targets greater than 13 g/dL differed from those in patients assigned to lower targets. None of the trials showed a benefit of higher Hb targets for these outcomes. Similarly, there is no evidence from the trials performed to date that higher Hb targets have a beneficial effect on left ventricular dimensions. With the exception of 1 small trial,6 RCTs also failed to show a benefit of higher Hb targets in terms of reducing the progression of kidney disease.
In developing the statement that in dialysis and nondialysis patients with CKD receiving ESA therapy, the Hb target should not be greater than 13.0 g/dL, the Work Group considered a meta-analysis performed by the ERT. The meta-analysis included published trials that reported results of all-cause mortality and adverse cardiovascular events in patients assigned to higher compared with lower Hb targets.
In patients with nondialysis CKD (predominantly stages 3 and 4), combining mortality outcomes from 8 studies with 3,038 individuals yields a risk ratio (RR) of 1.01 (95% confidence interval [CI], 0.63 to 1.61; Fig 2, left panel). Most deaths derive from the Correction of Hemoglobin and Outcomes in Renal Insufficiency (CHOIR)2 and Cardiovascular Risk Reduction by Early Anemia Treatment with Epoetin Beta (CREATE)1 studies, which together contribute 87% of the weight. Ordering studies chronologically in cumulative meta-analysis (Fig 2, right panel) shows that an earlier (1994 to 2005) nonsignificant trend favoring higher Hb targets resolves to a point estimate of 1 after addition of the 2 later largest studies.
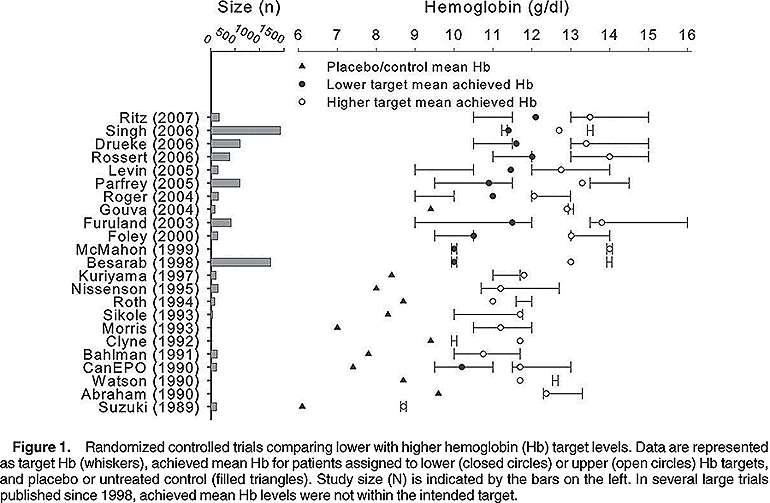
In patients with nondialysis CKD (predominantly stages 3 to 4), combining adverse cardiovascular events from 6 studies in 2,850 individuals yields an RR of 1.24 (95% CI, 1.02 to 1.51; Fig 3; left panel). Again, most events derive from the primary composite outcomes of the CHOIR2 and CREATE1 studies, which include deaths from any cause as a first event. Together, these 2 studies contribute 94% of the weight in this meta-analysis. The cumulative meta-analysis (Fig 3; right panel) shows that with the addition of these 2 studies, the point estimate moves from favoring higher Hb targets to favoring control treatment, a finding that becomes statistically significant.
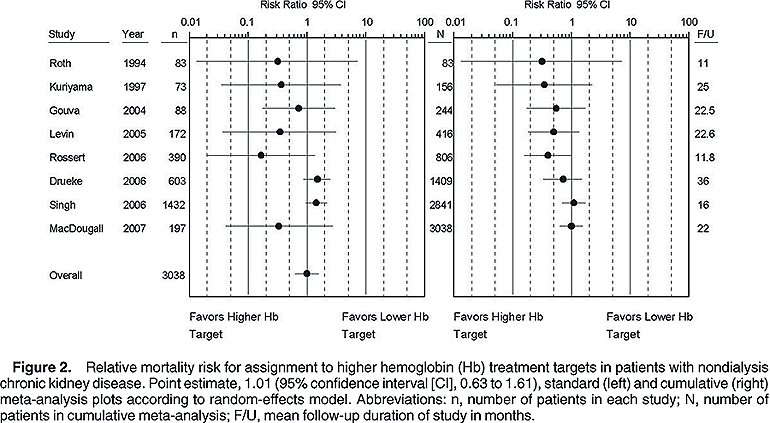
In dialysis patients with CKD, combining mortality outcomes from 4 studies with 2,391 individuals yields an RR of 1.12 (95% CI, 0.91 to 1.37; Fig 4; left panel). Most deaths derive from the study by Besarab et al,14 which contributes 81% of the weight.
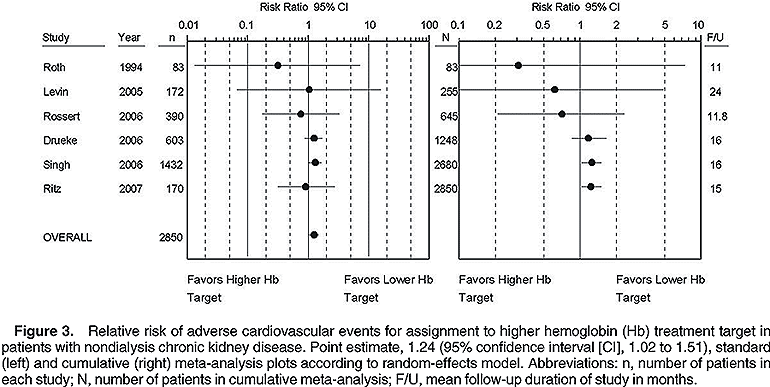
In dialysis patients with CKD, combining adverse cardiovascular events from 3 studies in 1,975 individuals yields an RR of 1.14 (95% CI, 0.79 to 1.64; Fig 5; left panel). Again, most events derive from the study by Besarab et al,14 which contributes 88% of the weight.
We compared both our methods and our results with those reported in another recent meta-analysis.28 We included RCTs with 6 months or longer follow-up without restriction on study size, whereas the previous metaanalysis included RCTs with 12 weeks or longer follow-up and greater than 100 subjects; our statistical model was more conservative (random- effects model always versus fixed-effects model if no statistical heterogeneity), and unlike the previous report, we did not pool studies in dialysis patients with those from nondialysis patients given the dissimilarities between these 2 target populations in ESA administration, Hb monitoring, and the presence or absence of dialysis. Finally, for cardiovascular outcomes, the previous meta-analysis included only myocardial infarctions, whereas we combined all cardiovascular disease events, including all events from the primary composite outcome in both CHOIR and CREATE. Thus, our definition of cardiovascular disease as an outcome was less precise, but more inclusive, than that of the other meta-analysis.
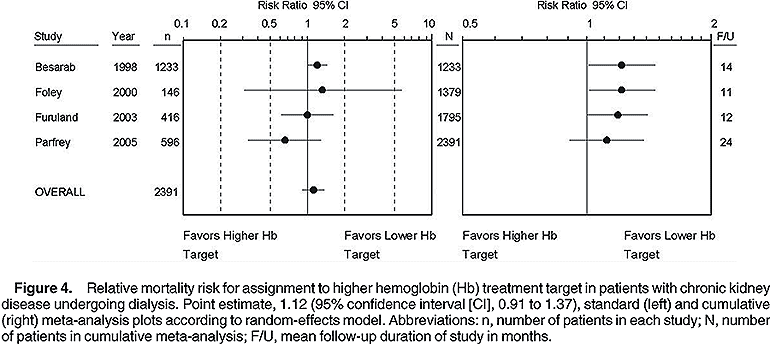
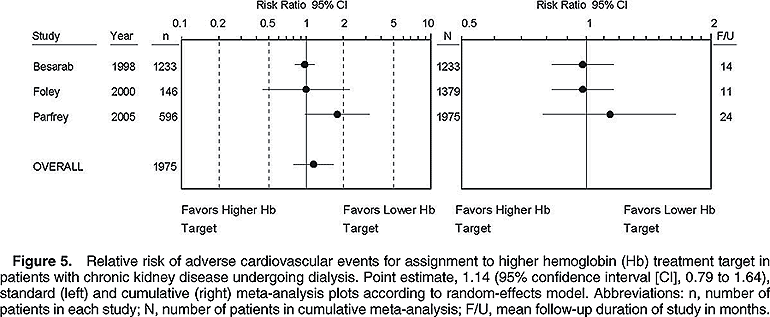
For mortality, our meta-analysis, like the recently published meta-analysis, showed no statistically significant difference for assignment to higher versus lower Hb level in either subgroup of dialysis or nondialysis patients. In nondialysis patients with CKD, we showed a RR closer to 1.0 and a wider CI (RR, 1.01; CI, 0.63 to 1.61 versus 1.33; CI, 0.98 to 1.81) than that previously reported because our analysis included results from 4 studies4,6,29,30 not included in the other meta-analysis. These 4 studies added 441 patients and 18 deaths (5 in the upper Hb arms and 13 in the lower Hb arms). In patients with CKD on dialysis, the 2 meta-analyses included the same studies and yielded essentially identical results (RR, 1.12; CI, 0.91 to 1.37 versus 1.11; CI, 0.94 to 1.31, current versus previous meta-analysis).
In appraising the overall evidence, the Work Group considered mortality, cardiovascular events, and HRQoL as outcomes of high importance. TheWork Group rated the evidence showing a trend toward greater cardiovascular events in dialysis and nondialysis patients assigned to Hb targets greater than 13.0 g/dL to be of moderately high quality for showing harm and of high quality for showing lack of benefit. The Work Group considered the HRQoL benefits in patients assigned to higher Hb targets as low quality evidence based on the limitations of reported HRQoL evidence (see the following section, Limitations of Evidence). The conclusion that in dialysis and nondialysis patients with CKD receiving ESA therapy, the Hb target should not be greater than 13.0 g/dL reflects the Work Group's judgment that the possibility to cause harm weighs more heavily than the potential to improve quality of life and to decrease transfusions.
The appraisal of the Work Group that the evidence for harm is moderately high renders statement 2.1.3 a moderately strong evidence based CPG. As discussed in more detail elsewhere (Methods), the designation moderately strong acknowledges the possibility that further research may alter either the appraisal of the quality of the evidence or the estimate of the effect size and thus result in a change in the guideline. The designation moderately strong therefore does not impede continued investigation.
Patient Outcomes
Most reports provide incomplete information with respect to HRQoL findings. Complete reporting should include point estimates and assessments of dispersion of HRQoL scores for each domain at each interval measured, by Hb target assignment.
Meta-analysis of cardiovascular events in dialysis patients is dominated by the results of the study by Besarab et al14 (1998), and in nondialysis patients with CKD, by the results of the study by Singh et al2 (2006). Although all RCTs have limitations, major limitations of those trials dominating meta-analysis results are of particular importance. In both Besarab et al14 (1998) and Singh et al2 (2006), the decision to prematurely stop the trial was made before the efficacy or futility boundaries were crossed. In Singh et al2 (2006), compared with the group assigned to the lower Hb treatment target, the group assigned to the higher Hb target showed at baseline a statistically greater proportion of patients with a history of hypertension and coronary artery bypass graft. A report posted by the study sponsor (PROCRIT ®: Clinical Study Report PR00-06-014 (CHOIR) SYNOPSIS, 12 September 2006; available at: www.clinicaltrials.gov, last accessed January 12, 2007) indicates that patients assigned to the higher Hb treatment arm also had a significantly greater severity of congestive heart failure (CHF) at baseline. The results of a multivariate analysis, included in this report, indicate that after adjustment for baseline conditions (CHF by National Health and Nutrition Examination Survey CHF score, atrial fibrillation/flutter, serum albumin level, reticulocyte count, and age), the relationship between treatment assignment and primary composite outcome events is no longer statistically significant (hazard ratio, 1.24; 95% CI, 0.95 to 1.62; P = 0.11 compared with the unadjusted hazard ratio of 1.34; 95% CI, 1.03 to 1.74; P = 0.03 reported in the publication2). Thus, although a trend toward greater risk of events in the higher Hb arm remains after adjustment for baseline imbalances, the finding of statistical significance is not robust and the change in the point estimate and CI with adjustment casts doubt on the success of randomization. Quality of the CHOIR study is further limited by censoring at the initiation of dialysis and by lack of information on when HRQoL was measured. One of the limitations of the CREATE trial is that the event rate was much lower than predicted; thus, the power to detect a difference in event rates was decreased.
Several studies are characterized by a failure to achieve the higher Hb target in the majority of patients at any time (Fig 1), and no study provided description of the Hb cycling around the achieved mean for either the higher or lower target treatment. In addition, several studies using subcutaneous (SC) epoetin alfa were prematurely terminated when reports of pure red cell aplasia emerged.
A further limitation of the currently available evidence is that important CKD subgroups have not been specifically studied or are not well represented in the existing studies, including children and young adults and patients with ischemic vascular disease or chronic lung disease.
Finally, trials published to date have not been designed to distinguish between the potential effects of Hb targets, ESA doses, and concomitant anemia therapy, including iron.
Implementation Issues
In clinical practice, medical decision making in the management of anemia at the level of the individual patient requires selection of the starting Hb level; choice of the initial dose, route, and frequency of ESA therapy; determination of Hb monitoring frequency; the aspiration to reach a threshold Hb or target Hb level; determination of the frequency and size of sequential ESA dose adjustments in relationship to a threshold Hb or target Hb level; and an interpretation of previous therapeutic trends and responsiveness to ESA therapy. Although available RCTs used either a range or a discrete value to represent the aspirational target Hb (Fig 1), published reports include little additional information to assist medical decision making. Specifically, information is lacking about how ESA and iron therapy were actually adjusted based on achieved Hb levels and how closely actual adjustments adhered to study protocol. Comparative information is similarly lacking to determine optimum frequency for monitoring Hb, the number of Hb results needed to reliably measure clinical performance, or the expected day-to-day within-patient variability in Hb levels in different patient populations (nondialysis CKD, hemodialysis CKD, and peritoneal dialysis CKD).
Aiming for a Hb target within narrow boundaries in ESA-treated patients requires frequent dose adjustments in many patients. More than 60% of patients receiving ESA therapy with Hb targets between 11.0 and 12.0 g/dL require between 6 and 9 dose changes per year.23 No comparative information is available to support evidence-based guidelines for the dosing and administration of ESAtherapy to achieve a target Hb. However, descriptive information from quality improvement interventions and RCT treatment protocols may be helpful to practitioners in weighing options that may best fit patient needs and practice settings.
In a 24-month study examining the effectiveness of a computer-assisted decision support algorithm for anemia management in hemodialysis patients, epoetin therapy (administered SC thrice weekly) was adjusted monthly in response to monthly Hb determinations by using stepwise ESA dosing adjustments, a lower Hb threshold below which ESA doses were increased, and an upper Hb threshold above which ESA downward adjustments were made.31 Epoetin doses were adjusted upward by 1,000 U/dose to achieve threshold Hb levels greater than 11.0 g/dL and downward by 1,000 U/dose once a month when Hb results exceeded predetermined ceilings (12.0, 13.0, and then 12.0 g/dL at intervals during the study) or by 50% if the Hb level exceeded 15.5 g/dL. In patients receiving epoetin doses near the mean for the study population (ranging from 9,800 to 6,400 IU/wk during the course of the study), a stepwise increase or decrease of 1,000 IU/dose represented on average a 10% to 16% change in epoetin dose. Although the ceiling Hb level alternated in 3 time periods between 12.0 and 13.0 g/dL, median achieved Hb remained stable, as did Hb variability around the median, with approximately 50% of achieved Hb results within 1.0 g/dL greater and less than the median at each monthly interval.31 A similar algorithm was used to adjust epoetin and darbepoetin doses given SC weekly.32
In hemodialysis patients, withholding ESA doses for a Hb level greater than target range is associated with subsequent downward Hb excursions, 23 often less than target range, consistent with the biology of erythropoietin as a cell salvage agent.56 The time between holding ESA doses and return of Hb to target range is variable and unpredictable. In hemodialysis patients with Hb values greater than 14.0 g/dL, the median time for Hb to return to 12.0 g/dL or less after holding of a SC-administered ESA is 7 weeks for long-acting ESAs (range, 2 to 13 weeks) and 9 weeks for short-acting ESAs (range, 6 to 13 weeks); the difference between long and shortacting ESAs is not significant.33
The effect of initiating a fixed monthly downward epoetin dose adjustment in response to achieved Hb levels greater than 13.0 g/dL was recently examined using the database of a large US dialysis provider.22 At baseline, approximately 35% of 95,000 patients receiving epoetin therapy showed average 3-month Hb results within the target range of 11 to 12 g/dL, and 15% showed average 3-month Hb results greater than 13.0 g/dL. When a computer-mandated 25% monthly dose decrease was initiated for end-ofmonth Hb results greater than 13.0 g/dL, percentages of patients with Hb values less than 11.0 and greater than 13.0 g/dL both increased. However, mean Hb level did not change.
The necessary ESA dose adjustment frequency may differ between initiation and maintenance of ESA therapy. In a randomized doubleblind trial comparing a short-acting ESA with a long-acting ESA in hemodialysis patients previously receiving epoetin alfa, dose adjustments were made in 25% increments or decrements of the baseline dose, aiming to maintain individual Hb concentrations within 1.0 and 1.5 g/dL of their baseline values and within a range of 9.0 to 13.0 g/dL. 34 Approximately 70% of patients required dose adjustment in the 20-week titration period, and 50% required dose adjustment during the 8-week maintenance period. Both dose increases and dose decreases were required. No between-group differences were seen in frequency or direction of ESA dose change.
Taken together, these reports suggest that when the target Hb level is 11.0 to 12.0 g/dL, variability of achieved Hb levels around the target is high, the fraction of prevalent patients with achieved Hb levels within the target range is low, ESA dose titration is required frequently during maintenance therapy, and either 25% ESA dose changes34 or 10% to 16% dose changes can be an effective maintenance dose-titration strategy.32 Clinical evidence is lacking about how to respond to achieved Hb levels greater than target range. Holding ESA doses may lead to steep downward Hb excursions and high amplitude Hb cycling.23 On the other hand, flexibility in determining the size of the dose adjustment may be needed. Imposing a fixed 25% dose decrease in response to greater-than-target Hb levels appears to promote greater Hb variability and more greater-than-target Hb values in patients undergoing maintenance therapy.22
In practice, when the target Hb is 11 g/dL or greater or 11.0 to 12.0 g/dL, achievement of Hb levels greater than the 11.0-g/dL threshold or greater than the 11.0- to 12.0-g/dL range is associated with lower mortality and less frequent hospitalization rates compared with achievement of lower Hb levels, an observation that is consistent in prospective longitudinal cohort studies16,18 and cross-sectional studies of large medical databases.17,19,20 In patients treated in facilities with the same target Hb, associated mortality and hospitalization rates are 10% to 12% lower for every 1.0-g/dL greater facility mean achieved Hb level.16 The same inverse relationship between achieved Hb and mortality and hospitalization also was seen in RCTs within assigned treatment arms (including arms with targets ≥ 13 g/dL).12,14 Failure to show between-group benefits in mortality, hospitalization, and left ventricular hypertrophy in patients assigned to higher compared with lower Hb targets in RCTs (Tables 2 and 8) confirms that the relationship between higher achieved Hb and lower risk is not causal. However, the results are consistent with the conclusion that when the target Hb is greater than 11.0 g/dL or 11.0 to 12.0 g/dL, achieved Hb levels greater than the 11.0-g/dL threshold or greater than the 11.0- to 12.0-g/dL range, whether facility specific or patient specific, do not constitute increased risk to patients. In general, higher facility-specific and country-specific achieved mean Hb levels and a lower percentage of patients with achieved Hb less than target range are associated with increased ESA use.16,25 However, adjustment for dose of ESA administered does not diminish the relationship between baseline Hb and mortality or hospitalization risk.16
Additional practical information on the use of ESA therapy to manage anemia in patients with CKD is provided in Section 3.1 of the KDOQI CPGs and CPRs for Anemia in CKD, published in May 2006.56
Measuring Performance
In general, a Hb target range suggests that ESA dose adjustment decisions are made by comparing the patient's achieved Hb with the selected Hb target. Although performance in managing to a Hb target can be expressed as the proportion of patients with Hb levels within the target range, in practice, only 30% of patients at any 1 time have an actual Hb level in the Hb target range of 11.0 and 12.0 g/dL when targeted to that range. The result of a single sampling in a single patient cannot be expected to lie within a narrow Hb target range (eg, Hb of 11.0 to 12.0 g/dL) or to equal a discrete point Hb target (eg, Hb of either 11.0, 11.5, or 12.0 g/dL). However, mean or median Hb levels of a group of patients or mean Hb levels of a single patient repeated over time would be expected to lie within a Hb target range or to approximate a discrete Hb target. In short, measures of clinical performance, to be clinically useful, must account for a high degree of within-patient and between patient variability.